Challenge: Visualizing extracellular vesicles
Extracellular vesicles (EVs) are key transporters of molecules, including both proteins and genetic material, from one cell to another. They are released from both healthy and disease-affected cells, and can carry harmful cargo such as viruses, toxins, neurodegenerative disease-associated, or cancer-linked proteins and RNA molecules when released from tumor cells (Candelario and Steindler). In this manner, EVs may be strong contributors to disease progression. Visualizing the interaction of EVs with target cells in real-time, in addition to tracking their movements within cells, may present important clues to their involvement in different disease pathways.
Extracellular vesicles are heterogeneous in size with the smallest sub-category being approximately between 30-120 nm in diameter. This means that in order to visualize them clearly, a resolving power beyond the capabilities of conventional light microscopy is needed. Additionally, while electron microscopy techniques have extremely high resolving power, they are limited to fixed cells and tissues, rendering them useless for tracking EV movements in real-time and within live cells.
Solution with the Nanoimager: Multi-method approach to understanding extracellular vesicles
 The Nanoimager is capable of a diverse range of imaging capabilities, allowing a combination of approaches to be taken in the investigation of extracellular vesicle dynamics and interactions. With whole body heating and a unique auto-focus system, the microscope is well adapted to perform live-cell imaging and trace the movements of EVs within cells and in solution, while maintaining perfect focus throughout long acquisitions.
The Nanoimager is capable of a diverse range of imaging capabilities, allowing a combination of approaches to be taken in the investigation of extracellular vesicle dynamics and interactions. With whole body heating and a unique auto-focus system, the microscope is well adapted to perform live-cell imaging and trace the movements of EVs within cells and in solution, while maintaining perfect focus throughout long acquisitions.
By following the trajectories of fluorescently labelled EVs in solution, the Nanoimager can apply nanoparticle tracking analysis (NTA) style sizing and quantification of vesicle populations. It also uniquely offers the ability to size EV populations in addition to visualizing and tracing their dynamics in a cellular context. The case study below illustrates an excellent example of this multi-method approach to understanding the role of EVs in cancer.
Case Study: Visualizing EV dynamics and interactions in living cells
 In this case study, the movements of an extracellular vesicle population, initially sized by NTA characterization, were visualized within cells post-internalization to investigate any interaction with the cell nucleus, as presented in the figure. The ultimate goal was to shed some light on the role of EVs in cancer.
In this case study, the movements of an extracellular vesicle population, initially sized by NTA characterization, were visualized within cells post-internalization to investigate any interaction with the cell nucleus, as presented in the figure. The ultimate goal was to shed some light on the role of EVs in cancer.
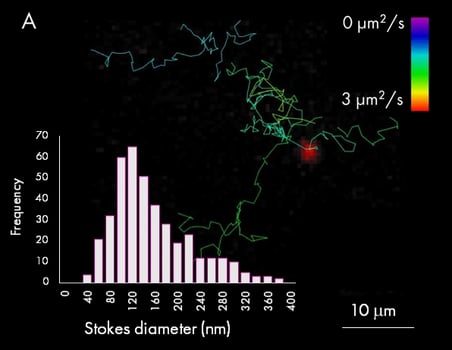
First, extracellular vesicles were purified from human Burkitt’s lymphoma cell line, labelled with AlexaFluor647 dye and purified by size exclusion chromatography. In this particular case, the automated particle detection and sizing feature was used to quantify and size the vesicle population in solution (panel A). The accompanying chart presents the size distribution of purified EVs, which peak at around 120 nm. The figure also shows example tracks colored by diffusion coefficients and a wide-field image of a single EV from one frame during a 1000 frames acquisition (red signal).
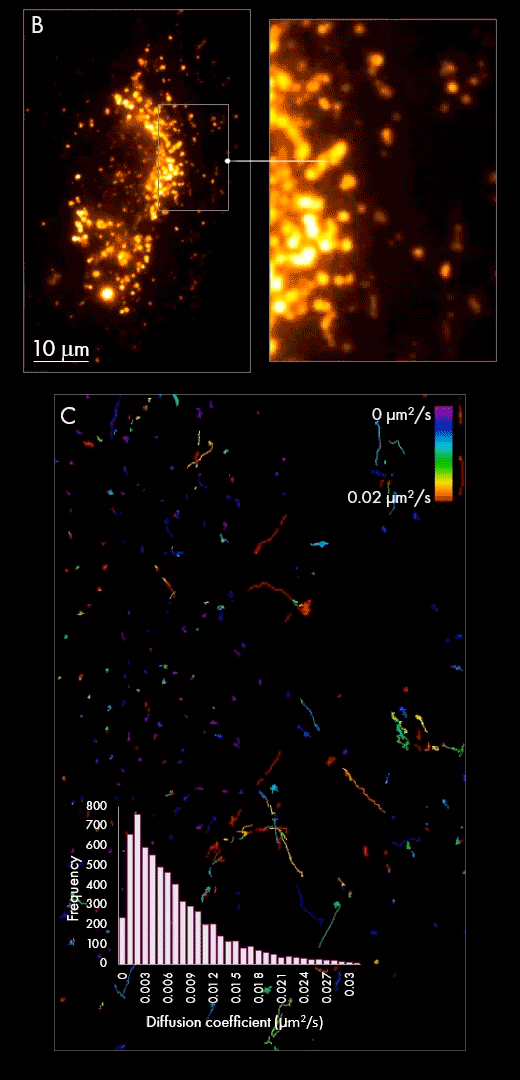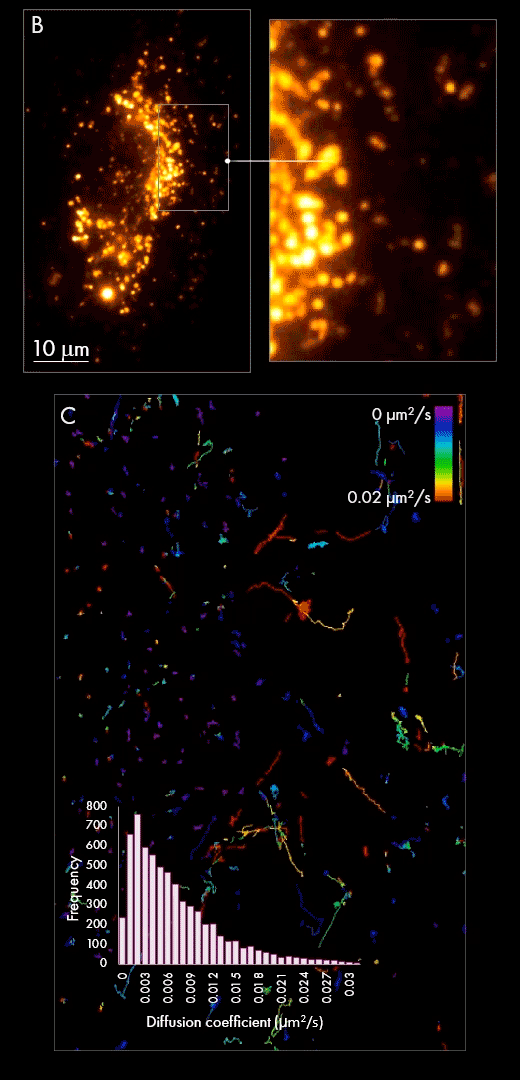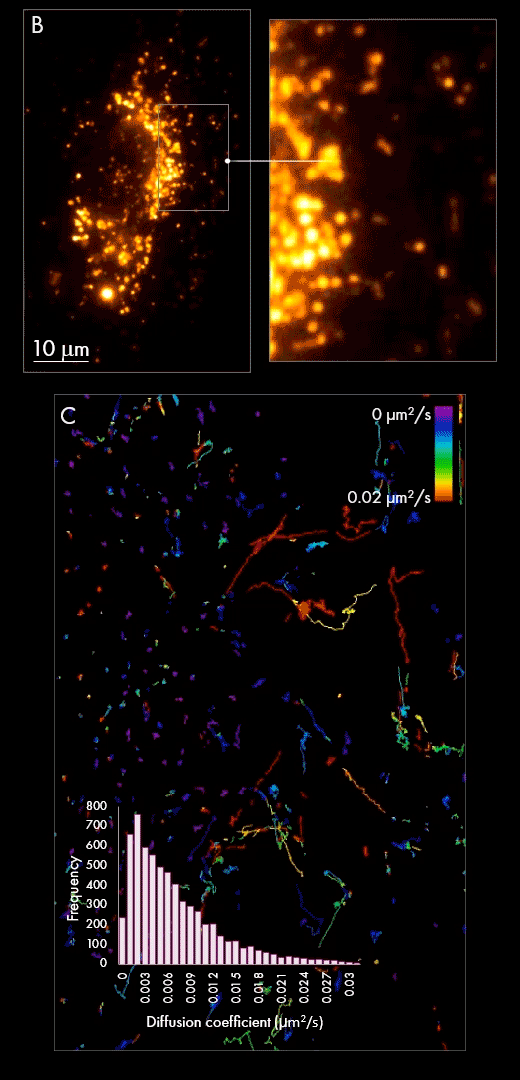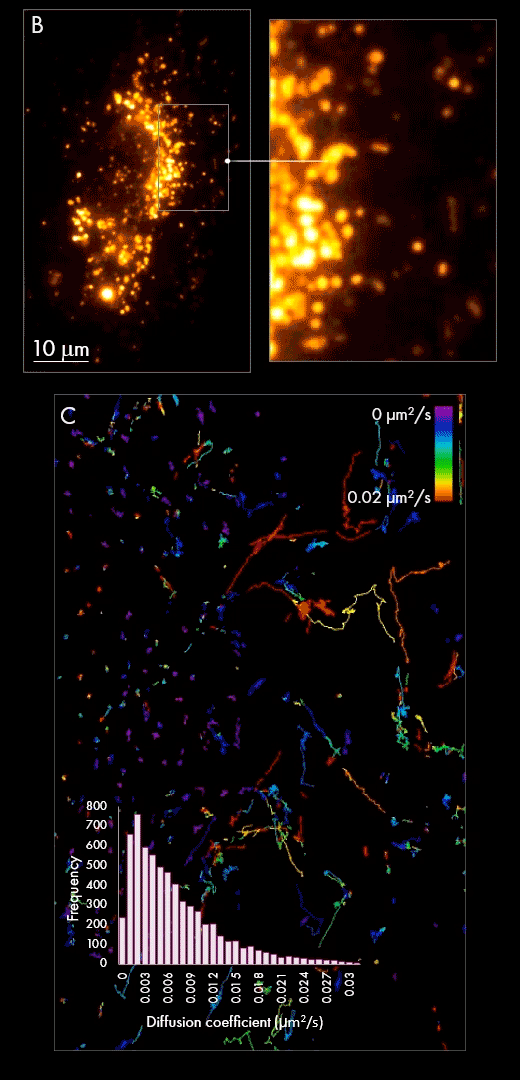
Secondly, purified populations of vesicles were incubated with cultured cells for several hours, giving them sufficient time to be internalized within the cell. Using localization-based single-particle tracking, the Nanoimager was used to image and trace the movement of EVs within the cell post-internalization (panel B). Track generation (an example is shown in panel C) indicated vesicle dynamics were slower in the area surrounding the nucleus in comparison to the cell periphery, as measured by diffusion coefficient.
Samples from Prof. C. Gregory Lab, University of Edinburgh
Read more about super-resolution microscopy of extracellular vesicles or its other applications such as vesicle and single-particle tracking in cells and viral particle imaging.
Share this article: